 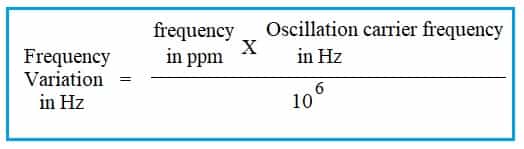
To calculate the PPM of one solid mixed with another, compare the masses of the two. That is, ppm = (mass of solute ÷ mass of solution) x 1,000,000. 
The concentration in parts per million or ppm is very similar to the weight percentage, except it multiplies the mass ratio by 1,000,000 instead of 100. How do you calculate the concentration of ppm? #How to calculate ppm how to#How to convert a number from percent (%) to parts per million (ppm). This number is then multiplied by 106 and expressed in parts per million (ppm). To find parts per million, divide the mass of the solute by the total mass of the solution. What is the formula for parts per million? Divide the mass of the solute by the total mass of the solution. #How to calculate ppm plus#The total mass of the solution is equal to the mass of the solvent plus the mass of the solute. Find the total mass of the solution in g. Measure the mass of the solute you want to mix with your solution. Method 2 of 3: Find the concentration in percent or parts per million. How they can calculate the concentration in ppm? So one part per million is a percentage: 1 ppm = To convert ppm to percent, divide ppm by 10,000: x (%) = x (ppm) / 10,000. How to Convert PPM to Percentage How to Convert Parts Per Million (ppm) to Percentage (%). You can either leave the measurement in milliliters or convert it back to liters to get 2.002 L.What does 1 ppm equal? One ppm is equal to 1 milligram of something per liter of water (mg/L) or 1 milligram of something per kilogram of soil (mg/kg). Add the volume of the solute to the volume of your solvent, ma.Then divide the each side by 2.16 to find the volume, or V = (3.45 g)/(2.16 g/mL) = 1.60 mL. Multiply each side by V to get V(2.16 g/mL) = 3.45 g. The formula would read 2.16 g/mL = (3.45 g)/V. In this case, the density of salt is 2.16 g/mL. Look up the density of salt either in a textbook or online and solve the formula for m. For example, if you’re finding the concentration of 3.45 grams of salt in 2 liters of water, you would find the volume of salt using the density formula.If you aren’t measuring the volume yourself, you may need to convert the mass of the solute into volume using the density formula.Measure the volume from the curve at the top of the solution, or the meniscus, to get the most accurate reading. 
If you’re finding the volume in a lab, mix the solution in a graduated cylinder or beaker and look at the measurement. The total volume of the solution is the amount of solvent plus the amount of solute added to it. This article has been viewed 1,848,785 times. This article received 14 testimonials and 81% of readers who voted found it helpful, earning it our reader-approved status. 
WikiHow marks an article as reader-approved once it receives enough positive feedback. There are 8 references cited in this article, which can be found at the bottom of the page. He also taught physical and life sciences and served as a teacher and administrator at public schools in California, Oregon, and Arizona. Hasegawa conducted biochemical research in Neuropharmacology at the National Institute of Health. He earned his PhD in Curriculum and Instruction from The University of Oregon. He holds a BS in Biochemistry, a Master’s in Education, and his teaching credential from The University of California, Davis. Hasegawa specializes in teaching complex scientific concepts to students. Chris Hasegawa was a Science Professor and the Dean at California State University Monterey Bay. This article was co-authored by Chris Hasegawa, PhD and by wikiHow staff writer, Hunter Rising. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
